Making Matches
 This is an extended version of one of my Popular Science columns: It contains more details and photographs than the magazine version. This version is not officially associated with the magazine: Responsibility for the contents is solely mine. This is an extended version of one of my Popular Science columns: It contains more details and photographs than the magazine version. This version is not officially associated with the magazine: Responsibility for the contents is solely mine.
Click any picture to open a larger version in a separate window.
I started this project with the goal of making a batch of functional matches using a minimum of complicated chemicals. The formula for commercial matches includes a bunch of things like antimony sulfide, which may improve some particular aspect of the match's performance (shelf life, manufacturing ease, etc), but which can in fact be omitted entirely without losing the basic functions of the match.
When you get right down to it, you can make a match using only three ingredients: Potassium chlorate, red phosphorus and Elmer's glue.
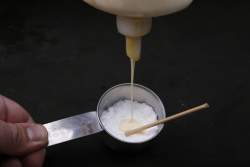 I started by mixing up a stiff paste of Elmer's yellow wood glue and potassium chlorate. I started by mixing up a stiff paste of Elmer's yellow wood glue and potassium chlorate.
 This mixture is fairly inert when wet: I've tried lighting it with a blow torch and it barely sustained combustion on its own. This mixture is fairly inert when wet: I've tried lighting it with a blow torch and it barely sustained combustion on its own.
 I rolled the mixture onto the ends of some 1/8" wooden dowels: It has to be a really thick mixture, otherwise it just sticks to the surface. (And of course you don't have to use a mini-anvil: The first time I just used the handle of a spoon, but the mini-anvil looked better for the magazine photos.) I rolled the mixture onto the ends of some 1/8" wooden dowels: It has to be a really thick mixture, otherwise it just sticks to the surface. (And of course you don't have to use a mini-anvil: The first time I just used the handle of a spoon, but the mini-anvil looked better for the magazine photos.)
 Then I baked the matches for a few hours in a warm but not hot oven, to dry the glue all the way through. How long it actually takes depends on how thick the match heads are: Mine were pretty big. As long as you keep the oven temperature in the warm-but-not-hot range (I didn't pay attention to the exact temperature I used) the matches are not likely to go off spontaneously. But I don't know where the ignition point is, and you should operate on the principle that they could catch fire in the oven at any point. With most ovens this wouldn't really be a safety issue since ovens are designed to get hot, but it could make a mess. Then I baked the matches for a few hours in a warm but not hot oven, to dry the glue all the way through. How long it actually takes depends on how thick the match heads are: Mine were pretty big. As long as you keep the oven temperature in the warm-but-not-hot range (I didn't pay attention to the exact temperature I used) the matches are not likely to go off spontaneously. But I don't know where the ignition point is, and you should operate on the principle that they could catch fire in the oven at any point. With most ovens this wouldn't really be a safety issue since ovens are designed to get hot, but it could make a mess.
 After drying, the mixture burns aggressively, pretty much like you'd expect a match head to. This picture shows one of these half-finished matches after it was lit with a torch. This is great so far, but a match you have to light with a torch isn't much good in the long run. After drying, the mixture burns aggressively, pretty much like you'd expect a match head to. This picture shows one of these half-finished matches after it was lit with a torch. This is great so far, but a match you have to light with a torch isn't much good in the long run.
 To create a real match, I mixed up a similar paste, not quite as thick, of Elmer's glue and red phosphorus, then dipped the tip of the dried matches in it one at a time. To create a real match, I mixed up a similar paste, not quite as thick, of Elmer's glue and red phosphorus, then dipped the tip of the dried matches in it one at a time.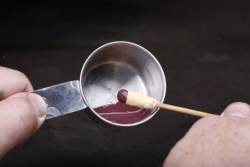
 To bake these dry, I stuck them in a cup full of sand, so the tips would stay nice and round. This second round of baking doesn't need to be as long since it's just a thin coating of red over the white paste. But they are also a good bit more likely to go off accidentally now that the red phosphorus has been added. To bake these dry, I stuck them in a cup full of sand, so the tips would stay nice and round. This second round of baking doesn't need to be as long since it's just a thin coating of red over the white paste. But they are also a good bit more likely to go off accidentally now that the red phosphorus has been added.
 As you can see from the picture, when you strike the match on some rough sandpaper, fire starts right at the boundary between the red and white pastes. What supposedly is happening is that friction from rubbing the red phosphorus on the sandpaper heats the red phosphorus, in microscopic areas, hot enough to convert it into white phosphorus, an extremely volatile and reactive substance. As you can see from the picture, when you strike the match on some rough sandpaper, fire starts right at the boundary between the red and white pastes. What supposedly is happening is that friction from rubbing the red phosphorus on the sandpaper heats the red phosphorus, in microscopic areas, hot enough to convert it into white phosphorus, an extremely volatile and reactive substance.
 The white phosphorus reacts with the potassium chlorate, releasing enough energy to set the potassium chlorate/Elmer's glue mixture on fire. After that, it's mainly Elmer's glue being burned by potassium chlorate: The red phosphorus did its job in the first fraction of a second of striking the match, and it takes only a tiny amount. In fact, in "safety matches" the red phosphorus is contained only in the striking surface, not in the match head. The small amount in the surface is enough to light all the matches in the box. The white phosphorus reacts with the potassium chlorate, releasing enough energy to set the potassium chlorate/Elmer's glue mixture on fire. After that, it's mainly Elmer's glue being burned by potassium chlorate: The red phosphorus did its job in the first fraction of a second of striking the match, and it takes only a tiny amount. In fact, in "safety matches" the red phosphorus is contained only in the striking surface, not in the match head. The small amount in the surface is enough to light all the matches in the box.
The matches I made actually worked better than I expected: Every one I tried lit after just a few strikes, which is more than you can say about some commercial matches! But there's a delicate balance between easy-to-light and too-dangerous-to-have-around. I did not save any of the matches I made, disposing of all of them by lighting them. I would not want to have them around because I do not have confidence that they would not, some day somehow, catch fire spontaneously. There may be some odd reaction that occurs with the Elmer's glue, who knows, and I for one don't want to find out.
One reason for caution is that the two serious ingredients, red phosphorus and potassium chlorate, are a very nasty pair when mixed directly. In these matches the Elmer's glue separates and tames the two ingredients. And when they combine to light the match, the Elmer's glue greatly slows down the rate of reaction.
But leave out the Elmer's glue and mix red phosphorus and potassium chlorate directly, and you have what is widely regarded as one of the most dangerous contact-sensitive explosive mixtures of all time. When I first read about it, my immediate reaction was of course to try it, but fortunately I read further before it was too late.
It turns out I even have a personal friend who has large scars on both hands from this stuff: Some forty years after he tried making it as a kid, he still sometimes sees a glint of light on his cheek while shaving. This is another sliver of test tube glass finally working it way out of his skin.
What makes this mixture so extraordinarily dangerous? The fact that in all likelihood it will explode while you are mixing it, not after it's finished and you've deployed it in some intentional way. As soon as powders of red phosphorus and potassium chlorate are brought in contact with each other, they may explode at any moment, for pretty much any reason: There are reports of such a mixture going of when it wasn't even being touched. Much worse is that it usually goes off while the inexperienced person, often a young one, has a jar or test tube of it in one hand and is mixing it with the other. The hand holding the test tube does not survive, while the one holding the stirring rod may only have the flesh torn off the fingers, depending on the amount of substance being mixed.
My friend Sherwin Gooch, of the glass slivers, has an interesting take on what lead him to his potentially fatal mistake: An excess of safety.I don't mind being used as a warning to others -- especially if it may prevent a younger someone like me from losing a hand (or worse).
I would like to relate what I call the "Gilbert Chemistry Set Effect". I had a chemistry set. I did all the experiments in the book, then mixed together pretty much every combination of chemicals I could think of...and pretty much nothing happened. Some bubbles, some color changes. Then I got my hands on an industrial chemical supply catalog. I WAS IN THE HOSPITAL WITHIN TWO DAYS OF RECEIVING MY FIRST ORDER! And I was lucky I didn't take most of my family with me.
Sometimes things can be too safe. The Gilbert Chemistry Set fostered a sense of false security. It caused me to become blase about what chemicals can do. That's what I call the Gilbert Chemistry Set Effect.
Sherwin It goes without saying that I of course did exactly the same thing with my Gilbert Chemistry Set. I was lucky: Some of the things I did with flash powder rockets were truly stupid, but I never got hurt. And Sherwin was lucky too: His hands have some scars, but they work just fine. Many other people have not been so lucky, and it is a constant worry of mine (and my editor's lawyer's) that something I write about may lead someone, probably a teenager, to seriously injure themselves. Blinding is the most likely serious injury, and would certainly have been Sherwin's fate had he not been wearing glasses.
But Sherwin is right: Making things safer does not always lead to increased safety. We don't have kids spend years practicing with Nerf cars that don't hurt when you get run over by them, then one day just throw them out in the real world of real cars.
Kids are very smart, what they lack isn't the ability to understand danger, it's the judgement to determine on there own what is dangerous. Mix one pair of powders together and nothing happens. Mix another pair and you're blind for the rest of your life. Experienced chemists misjudge these things sometimes, teenagers certainly do too. No shame in that, but someone's got to tell them which of the powders to be careful with, because sooner or later one of them is going to try it.
The current fashion is to simply say everything is dangerous and you shouldn't try anything at home. This is a mistake. After the third or fourth time you've been told you have to wear safety glasses to mix up sugar water, the average kid, not to mention the average doorknob, figures out that this is not serious advice. It's crying wolf, and it's dangerous for exactly the same reason those sheep ended up as dinner.
In my Popular Science columns I try, within the constraints of the magazine's understandable liability concerns, to get people to understand which things really are dangerous, and which we're just saying you shouldn't do because the lawyers require it.
Having said all that, let me repeat: The mixture of red phosphorus and potassium chlorate is really really really dangerous. It is so dangerous you should not try to do it, period. Experienced pyro-technicians simply don't bother, because they know there are plenty of other ways to make explosives that work much better: Bigger bang, fewer mangled hands, more easily available ingredients.
Chemists do sometimes demonstrate this reaction, mainly to show how crazy it is. (In fact, the one time I've seen it done was by my untamed Icelandic chemist friend Tryggvi, who works with Sherwin.) They mix a tiny amount only, a thumbnail's worth of each at most, and they mix it with a feather or by shuffling it back and forth on a sheet of paper. They never, ever, put it into any kind of container. Containment greatly increases the explosive power of any substance, and this is no exception. (A container also provides material to be ejected at high velocity.) Placed on a flat surface, the mixture can be set off with a light tap of a long-handled hammer. It goes bang. It's not worth the risk.
Well, what does this say about matches? That they really are poised on the brink of mayhem. Historically matches were anything but safe, and quite a number of deaths were attributed to their going off when it was not wanted. (A no doubt much larger number can be attributed to them working as intended, but that's another story entirely.) Modern matches are an amazing achievement in balance: They inevitably must contain highly volatile, even explosive chemicals, otherwise there's no way they could light with nothing more than rubbing. But they must, and do, keep those chemicals reliably under control until just the right moment. This is chemistry of the highest order.
|
|